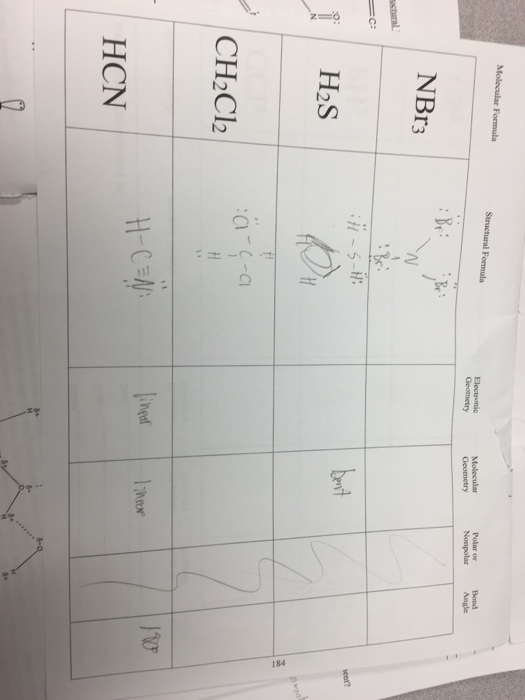
We separate this into two categories, the electron-group geometry and the molecular geometry.Įlectron-group geometry is determined by the number of electron groups. Although VSEPR theory predicts the distribution of the electrons, we have to take in consideration of the actual determinant of the molecular shape. Thus, the molecule's shape reflects its equilibrium state in which it has the lowest possible energy in the system. The electrons and the nuclei settle into positions that minimize repulsion and maximize attraction. The shape of a molecule is determined by the location of the nuclei and its electrons. Using the VSEPR theory, the electron bond pairs and lone pairs on the center atom will help us predict the shape of a molecule. An electron group can be an electron pair, a lone pair, a single unpaired electron, a double bond or a triple bond on the center atom. VSEPR focuses not only on electron pairs, but it also focus on electron groups as a whole. Thus, electron pairs will spread themselves as far from each other as possible to minimize repulsion. The valence-shell electron-pair repulsion (VSEPR) theory states that electron pairs repel each other whether or not they are in bond pairs or in lone pairs. Now that we have a background in the Lewis electron dot structure we can use it to locate the the valence electrons of the center atom. Valence-Shell Electron-Pair Repulsion Theory HCN is also used to prepare pesticides and chemical warfare agents.\( \newcommand\) It reacts with bases to form salts called cyanides.ĭespite its toxicity, HCN is a necessary reagent used in the production of a variety of useful industrial chemicals such as sodium cyanide, potassium cyanide, methyl methacrylate (monomer used for making polymers and plastics), chelating agents EDTA and NTA, as well as the polymer Nylon. Its chemical name is hydrogen cyanide, which is a weak acid, and partially ionizes in water to give H + (or H 3 O + ) and the cyanide anion, CN. It has a distinct smell of bitter almonds, which is used to identify the presence of this highly poisonous material. Hydrocyanic acid features a density of 0.687 g/mL, and boils slightly above temperature, at 25.6 ☌ (78.1 ☏). HCN is found as a pale blue, colourless transparent liquid (hydrocyanic acid) or a colourless gas (hydrogen cyanide). 
HCl + NaCN → HCN + NaCl HCN Physical Properties HCN is prepared on a laboratory scale by the addition of acids to cyanide salts of alkali metals (such as NaCN, KCN, etc.): HCN occurs naturally in the pits of certain fruits such as cherries, apples, and apricots, and the fruit pits contain small amounts of cyanohydrins from HCN. Hydrogen cyanide may be a simple planar molecule, with a triple bond between the carbon and nitrogen atoms. Its molecular formula is written as CHN, and its molar mass is 27.03 g/mol. The chemical formula of hydrocyanic acid is HCN. Organic compounds such as aldehydes and ketones, forming cyanohydrins, which function intermediates in many organic syntheses. The salts used in the extraction of ores, in electrolytic processes, and the treatment of steel. In pure form, the compound may be stable, but it polymerizes readily within the presence of essential substances, like ammonia or cyanide. Hydrogen cyanide is an excellent solvent for several salts, but not widely used as a solvent due to its toxicity. A method of inflicting execution consists of administering a deadly dose of the compound gas. Humans can survive 50–60 parts of the compound per million parts of air for an hour without severe costs, but exposure to concentrations of 200–500 parts per million of air for a half-hour is typically fatal. 
Hydrogen cyanide is very toxic because it inhibits cellular oxidative processes. It also is employed in the preparation of acrylonitrile, which is used in the production of acrylic fibres, synthetic rubber, and plastics. Hydrogen cyanide compounds are used for several chemical processes, including fumigation, hardening of iron and steel, electroplating. 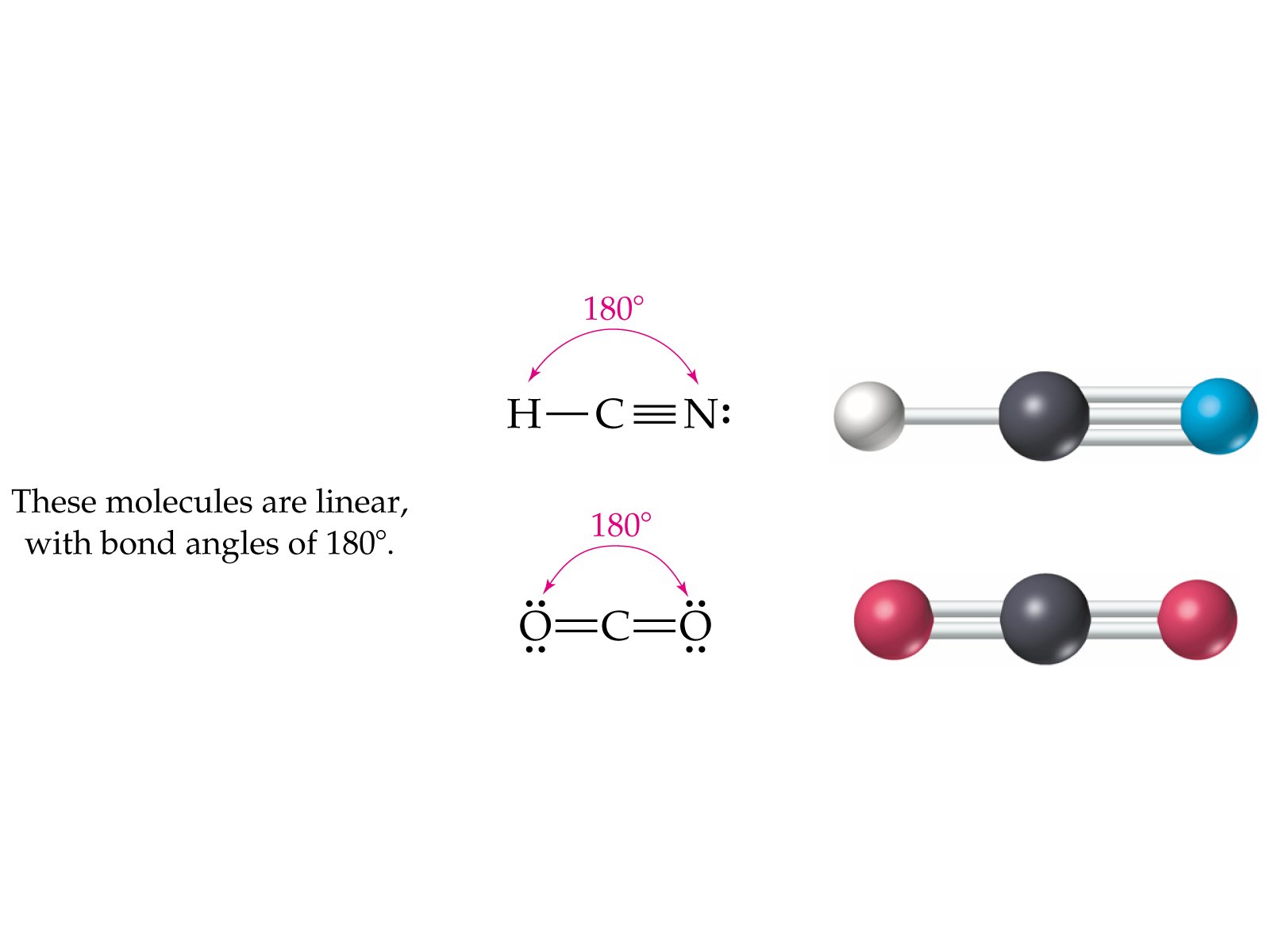
It was discovered in 1782 by a Swedish chemist, Carl Wilhelm Scheele, who prepared it from the pigment Prussian blue. A solution of the compound in water is named prussic acid. It has a boiling point 26° C and freezing point -14° C. HCN acid name is hydrogen cyanide, a highly volatile, colourless, and extremely poisonous liquid.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
